 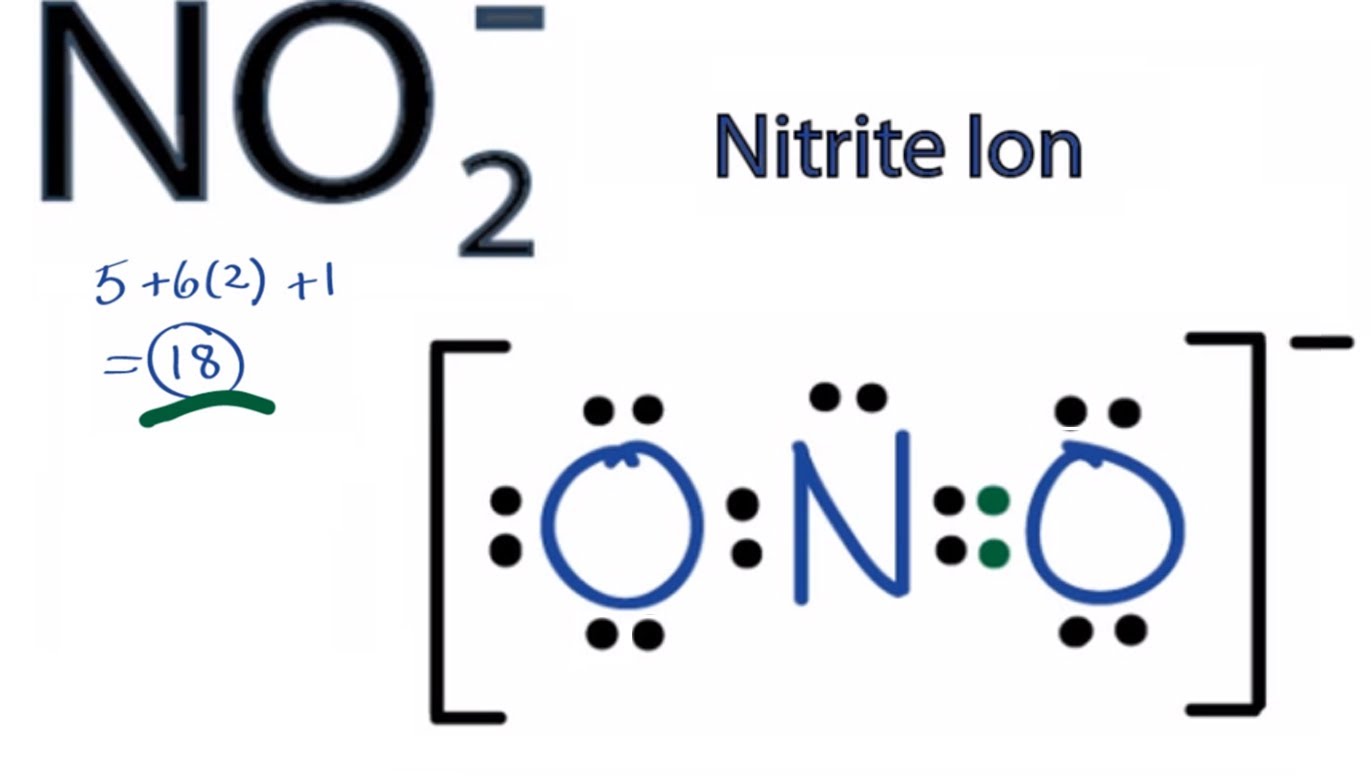
It does not fluctuate between resonance forms rather, the actual electronic structure is always the average of that shown by all resonance forms. The first part is the number of valence electrons the atom donates to the Lewis dot Structure. We can calculate an atom's formal charge using the equation FC VE - LPE - (BE), where VE the number of valence electrons on the free atom, LPE the number of lone pair electrons on the atom in the molecule, and BE the number of bonding (shared) electrons around the atom in the molecule. We should remember that a molecule described as a resonance hybrid never possesses an electronic structure described by either resonance form. Calculating Formal Charge: The following equation determines the formal charge for each atom in a molecule or polyatomic ion. 
We can use this information to calculate the formal charge on each atom in a. By subtracting one-half the number of bonding electrons, we essentially. Chemists use formal charges on atoms as one important piece of evidence in. Brackets surround this structure, and there is a superscripted negative sign. The formal charge is equal to the number of valence electrons. The right structure shows an oxygen atom with three lone pairs of electrons single bonded to a nitrogen atom with one lone pair of electrons that is double bonded to an oxygen atom with two lone pairs of electrons. 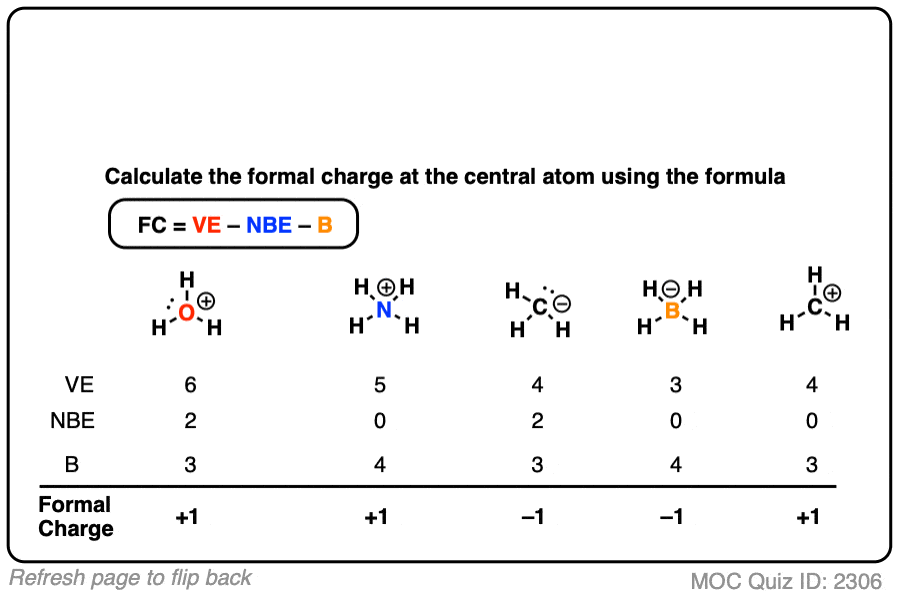
Brackets surround this structure, and there is a superscripted negative sign. One can calculate the formal charges for any given atom with the help of the following formula: F. These charges help in knowing if the given structure of the molecule is stable or not. The left structure shows an oxygen atom with two lone pairs of electrons double bonded to a nitrogen atom with one lone pair of electrons that is single bonded to an oxygen atom with three lone pairs of electrons. Formal charge is the individual electric charges on the atoms in a given polyatomic molecule. Two Lewis structures are shown with a double headed arrow drawn between them. The molecular mass calculator will recognize the entered formulas, which are included in the list of organic compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
